Ear Medications
Cortotic®
Treatment for canine otitis externa without an antimicrobial
Cortotic® is a corticosteroid-only first-line treatment for canine otitis externa without an antimicrobial.
- Proven to relieve pain and pruritus1
- Proven to control microbial overgrowth1
- As effective as a triple-active ear formulation1
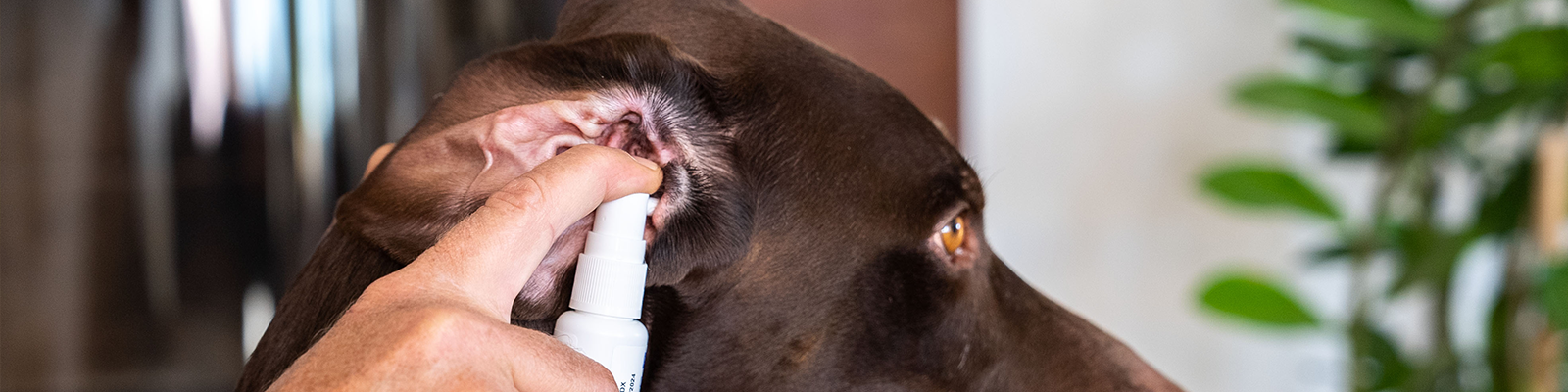
- Easy to use and gentle on ears2
WHAT IS CORTOTIC®?
Cortotic® contains the potent corticosteroid hydrocortisone aceponate (HCA), and is proven to treat the most common form of canine otitis externa*, without the need for antimicrobials**.
- The only corticosteroid licensed as a first-line treatment for acute erythemato-ceruminous otitis externa
- Antibiotic and antifungal-free: Proven reduction in bacterial and yeast overgrowth, without antimicrobials
- Proven efficacy: Rapid relief from clinical signs, including pruritus and pain
- Gentle application: Atraumatic rounded nozzle and fine mist for even application
- Dose controlled: 2 pumps per application whatever the dog's size. Administer just once per day, for 7-14 days.
DOSAGE AND ADMINISTRATION
Use as directed by prescribing veterinarian. Screw the pump spray onto the bottle and prime the pump by pressing it until the product is released. Hold the product upright while administering treatment. Introduce the atraumatic cannula into the ear canal. Administer two pump activations (0.44 ml) into each affected ear. Massage gently to ensure even distribution of the product. Avoid contact with the dog's eyes by restraining the dog's head to prevent shaking. In case of accidental contact, rinse thoroughly with water. Repeat treatment once daily for 7 consecutive days. Use the contents within 6 months of first broaching the container. Discard the unused portion.
STORAGE
Store below 25°C.
PACK SIZE
Available in 16 ml bottles.
Talk to your vet about Cortotic®.
Restricted Veterinary Medicine. Available only under Veterinary Authorisation. Registered pursuant to the ACVM Act 1997, No. A012028. See www.foodsafety.govt.nz for registration conditions.
*For the treatment of acute erythemato-ceruminous otitis externa.** A secondary reduction of bacterial and yeast overgrowth was demonstrated, and concomitant treatment with an antimicrobial was unnecessary.
This product is therefore recommended as a first-line treatment.
1. Rigaut D, Briantais P, Jasmin P, Bidaud A. Efficacy and safety of a hydrocortisone aceponate-containing ear spray solution in dogs with erythemato-ceruminous otitis externa: A randomised, multicentric, single-blinded, controlled trial. Vet Dermatol. 2024 Apr;35(2):197-206. 2. Pet-owners satisfaction of use questionnaire after the clinical field efficacy study.




